December 22, 2021
Logistic solutions for CAR T-cell therapy - let's scale!
In the past several years, we have witnessed a breakthrough for autologous CAR T-cell oncology immunotherapy with Novartis’ Kymriah (tisagenlecleucel) and Kite Pharma’s Yescarta (axicabtagene ciloleucel) being approved for treatment of refractory acute lymphoblastic leukemia (ALL) and relapsed large b-cell lymphoma by the U.S. Food and Drug Administration (FDA) in 2017. Worldwide, almost a thousand high potential cell and gene therapies (CGT) and allogeneic and autologous adoptive cell transfer therapies for pediatric and adult patients are in clinical trials right now. The massive scale-up required to successfully commercialize and provide these therapeutics to patients led to a paradigm shift for biotech companies and healthcare centers alike.
Strikingly, the biggest challenges are not on the medical or life science sides of the equation, but rather in the associated patient-centered and personalized logistics that are unprecedented in the pharma business.
As an example, the manufacturing process for chimeric antigen receptor T-cell therapy (CAR T-cell therapy) requires harvesting of the patient’s lymphocytes (T-cells) in a process termed apheresis. Next, the frozen cells need to be transported under cryogenic conditions to the manufacturing facility, which might be a trip across the globe. After modification and culturing of the cells under Good Manufacturing Practice (GMP), the cellular therapy must make its way back to the patient, while ensuring an uninterrupted chain of custody and chain of identity.
In a nutshell, for these highly advanced therapies, every single patient requires his personalized supply chain.
Fully personalized Supply chain
The bottleneck for CAR T-cell therapy and similar cell and gene therapies is the logistic complexity of orchestrating each cog in the machinery of the process. Biopharma companies need to develop partnerships to overcome this novel supply chain problems, which are unprecedented in traditional pharma business:
- predictable and short time scales for cell removal, modification, and transport to the patient.
- high modification success rate
- maximal efficacy and minimal adverse effects rates for all patients
- economic pricing models
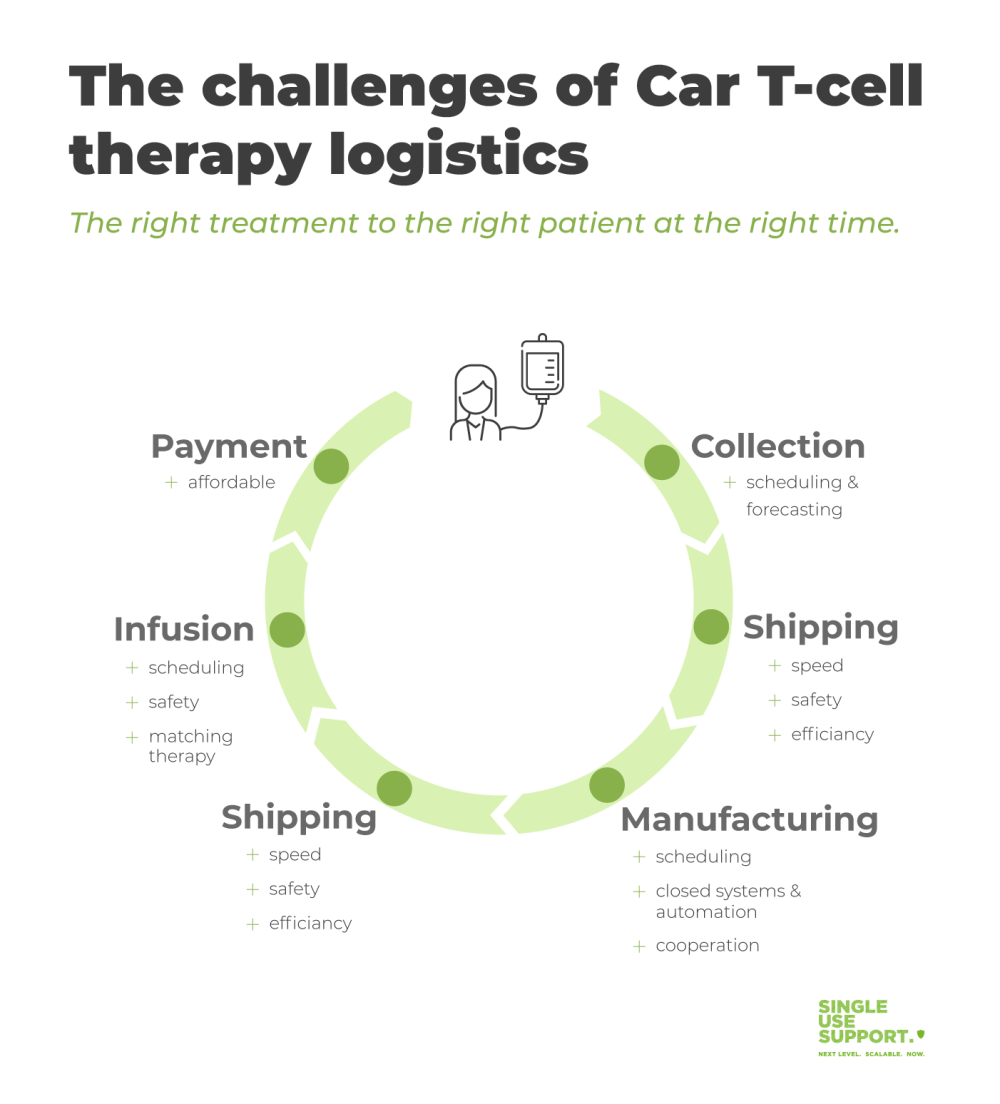
Those requirements lead to the following challenges:
- Delivery model: cell and gene therapy are made to order. Manufacture of treatments in advance to keep an inventory is not possible.
- Raw materials: are obtained from patients. This is the first step in the production process. Quality and viability of cellular material is highly variable with the patients’ history (age, immune system’s status, prior chemotherapy, etc.)
- Absence of scale: one batch per patient
- Unknown supply conditions: projections for demand and supply constraints are largely unknown yet and biopharma companies need to project for a wide variety of scenarios.
- Chain of custody and chain of identity: the individualized nature of these treatments is crucial for their effectivity. This translates to the absolute requirement that each patient receives her own CGT product to avoid devastating toxicity, enabled by consistent end-to-end monitoring and tracking of their therapy
Vein-to-vein workflow - the logistics challenges in CAR T-cell therapy
Reliable monitoring of several critical parameters during the vein-to-vein workflow is needed to guarantee the administration of effective cell products. The challenge is unifying data from at least three sources: clinicians at healthcare centers, the manufacturing unit and the logistics provider that transports the goods between them.
Often contract manufacturing organizations (CMOs) are involved during periods of high demand. Each institution has its own standard operating procedures (SOPs) and ways of storing and processing data. Therefore, real-time communication that works during comparably small clinical trials, is expected to collapse during massive scale up during commercialization.
Effective control of the process is only possible with a unified, central data collection and monitoring service provider.
The digital supply chain in CAR T-cell therapy logistics
All parts of the solution to this challenge are already available through recent IT developments and can be applied to solve the puzzle of the digital supply chain. Cloud storage and cloud computing will be major pillars in establishing cell and gene therapies as broadly available for cancer patients.
A central database, in which all stakeholders log their data and monitor the advancement of each individual therapy development, takes care of inter-institutional communication and allows for effective projection of production capacities and treatment windows. Additionally, it facilitates process optimization, and allows direct intervention in case of emergency.
Oftentimes, the implementations of such systems struggle with compatibility issues. Surprisingly, many culprits are hidden at a very basic level. As an example, standardized and compatible cell therapy manufacturing hardware is not as broadly used as needed.
Car t-cell therapy - packaging, freezing and protecting - the cold chain reality
The use of unstandardized hardware for critical steps during CGT manufacture is still a factor in the rate of loss-of-goods.
The recent COVID-19 pandemic has catalyzed the development of cryogenic supply chains and associated equipment for large volumes vaccines. These advances naturally spilled over to CAR T-cell therapy and other CGT approaches and sparked innovative solutions.
Single Use Support is a torchbearer in cryogenic drug substance logistics and our R&D department supports a team that is in constant interaction with market participants to develop scalable solutions and optimize processes for protecting, fill & freeze, shockfreezing, packaging and handling of small volume cell and gene therapeutics.
Download our APP NOTE on controlled cell filling & freezing
Discover how automated fluid management along with controlled-rate freezing can be used in cell-based biomanufacturing processes to improve cell viability and process safety.



