APPLICATIONS BY PROCESS

Advanced Fluid Management and Freeze/Thaw Logistics
We close the gap between downstream and the fill & finish process in the biopharma industry with the use of single-use technologies. At Single Use Support we offer biopharma companies access to a new and almost 100% secure drug substance logistics process for their filling, freezing, thawing and logistics processes.
Our developed technologies increase patient safety and minimize the risk of bio contamination and product loss significantly. We build upon standards, eliminate material deficiencies, and minimize human errors.
Storage and shipping of high-value substances such as APIs (active pharmaceutical ingredients), buffer, media, biojoncugates, vaccines, cells or other liquids can be performed faster and more safely than before.
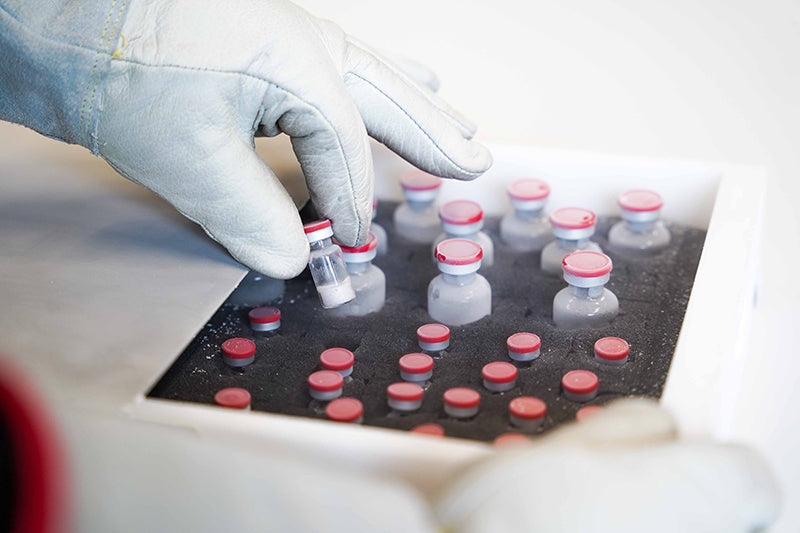
Cell Banking
In the process of creating new drug compounds cells play an important role. However, handling of cells requires very carefull and fast processes in order to guarantee best-possible quality.

Fermentation
Cold Chain Handling for Microbial, Bacterial & Mammalian Fermentation. For improved fermentation production, manufacturing suites must be equipped with scalable, cost-effective and robust process solutions for large volumes up to hundreds of liters per batch.

Fill/Filtration-Freeze/Thaw
cGMP-compliant single-use technologies enable standardization in critical process steps where a large technology gap exists in biopharmaceutical production.