Autologous vs. Allogeneic Cell Therapy
Table of contents
ShowComparing autologous vs allogeneic cell therapies, it becomes apparent that they share both defining methodological key elements as well as various fields of application, including most innovative cancer therapies, there are significant differences between them. The most important one is the origin of the transplanted cells.
While donor’s and patient’s body during an autologous stem cell therapy correspond, patients undergoing an allogeneic stem cell transplant receive cells from a donor other than themselves.
Their effects being shown in bone marrow transplantation by Dr. Donnall Thomas in the mid-20th-century, cell therapies since then have allowed huge leaps in the treatment of various cancer types, like leukemia or lymphoma, and several other diseases. As for today, numerous clinical trials have been carried out to maximize their effectiveness and reduce side effects, in that they nowadays represent well-established treatment options.
In order to get a more detailed overview on what the core differences, challenges and chances of these two approaches are, a closer look on the procedures of stem cell transplantation will be presented.
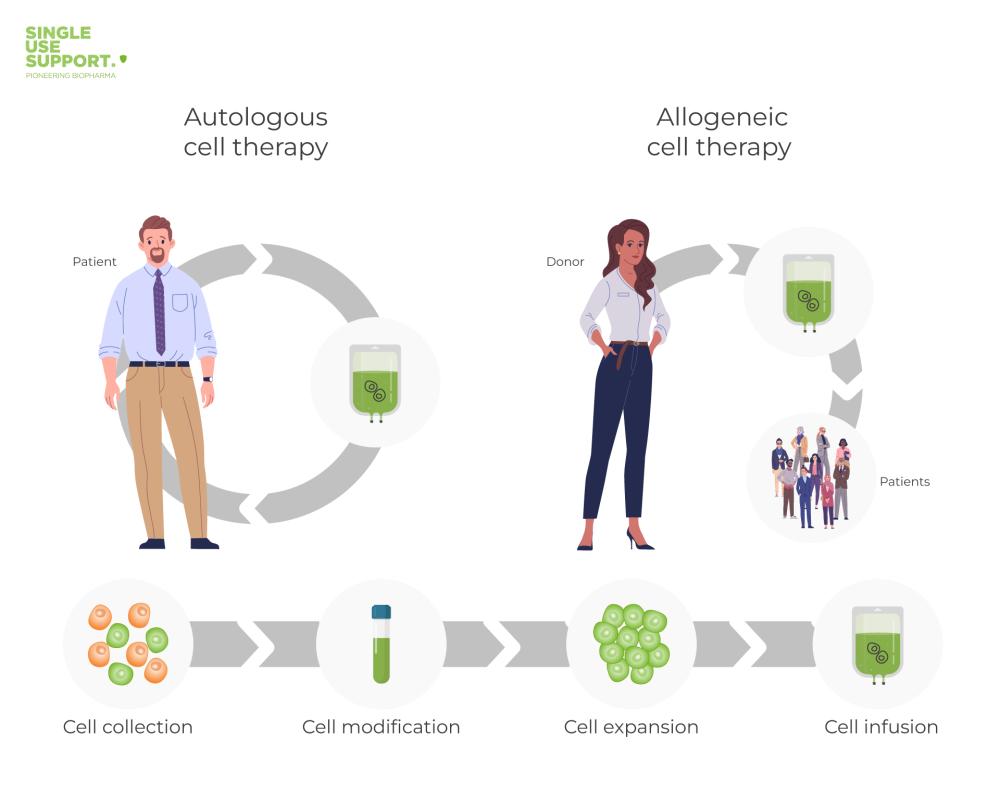
Autologous and Allogeneic Stem Cell Transplant: The difference in one image
Autologous Stem Cell Transplantation
Autologous cell therapy refers to a therapy using a patient own's cells: A patient’s own cells are extracted in order to further analyze the different cell types “ex vivo” (outside one’s body). Consequently, measures can be taken to treat and prepare cells for reinsertion. Their engraftment followed by their reproduction can be performed in order to replace unhealthy cells in the patient’s body, by the means of knowledge from various scientific fields (such as virology, oncology, immunology or hematology).
Equally large is the number of medical conditions for which autologous cell transplantation has proven to be a promising treatment option: It can, for instance, be taken into consideration for patients suffering from multiple myeloma or lymphoma, or be part of immunotherapy. Cartilage cells, on the other hand, have been successfully used to repair joint damages, thus underlining the huge potential of autologous cell therapy in regenerative medicine as well.
Further reading: What is autologous?
Allogeneic Stem Cell Transplantation
Allogeneic Therapy refers to a therapy using cells from a donor. As they are collected from one individual (this can also be an unrelated donor or a haploidentical donor) in order to be given to several patients, cells used in allogeneic therapy were not originally the recipient’s own. The collected donor cells might directly be given to a genetically similar and compliant patient or added to a cell bank. This is why these cell products are often considered to be offering an “off-the-shelf”-supply for many patients, as multiple batches can be produced out of a single donation followed by a complex manufacturing process.
Allogeneic stem cell therapy is frequently used for patients whose own stem cells are ineligible for a transplant, e.g. those affected by hematological malignancies like leukemia. Patients with blood cancer, for instance, would otherwise be at high-risk of recidivism, as cancer cells might be reinserted. Furthermore, an allogeneic stem cell transplant deriving from the bone marrow and blood of a healthy donor is crucial in curing myelodysplastic syndromes.
Further reading: What is allogeneic?
Autologous vs allogeneic cell therapies – what is the difference?
The main differences between autologous and allogeneic cell therapy coincide with the origin of the used cells. As already described, an allogeneic stem cell transplant derives from a healthy donor. Most commonly when performing a hematopoietic stem cell transplantation, the required cells are harvested in the donor’s tissue. Usually derived from peripheral blood, the bone marrow or umbilical cord blood, the targeted cells undergo a manufacturing process to gain batches for sometimes thousands of patients. Meanwhile, the cells used for an autologous stem cell therapy come from the patient’s body and are usually not considered as an intruder by one’s immune system, therefore not causing adverse events.
Consequently, an autologous stem cell transplant must be prepared exclusively for one recipient, being the donor at the same time. Nevertheless, this tailor-made treatment often cannot be performed when a patient’s own cells are affected by diseases like leukemia.
Challenges of autologous and allogeneic cell therapies
Considering an autologous stem cell transplant, one must be aware of several difficulties: A limiting factor might be to collect a sufficient number of cells suitable to undergo the following manufacturing process with a sufficient proliferation rate. Also, these cells are not very durable ex-vivo, in a sense that they need to be ready for reinsertion within a short period of time.
On the other hand, patients receiving an allogeneic stem cell transplant are at a higher risk of facing GVHD (graft-versus-host disease) in the follow-up time, when the donor’s transplant fights the patient’s body. Although examining the compatibility of donor and patient via HLA-classification prior to the procedure, this activation of an unwanted immune response cannot be excluded. Nevertheless, great progress has been made in the application of MSCs (mesenchymal stem cells), in that no additional immunosuppression is necessary. Another risk is regimen-related toxicity of organs.
Additionally, cell therapies in general and consequently also these two types of stem cell transplants have to deal with the immense sensitivity of the extracted cells alongside the risk of their contamination.
Single Use Support's solution
In the past few decades, cell and gene therapy has taken huge steps as an established treatment option for various medical conditions.
One promising approach in gene therapy, for instance, is Car-T cell therapy: A patient’s leukocytes (i.e. white blood cells) are modified and production of the chimeric antigen receptor (a protein that, similar to an antibody, detects antigens) is induced, programming them to fight specific cancer cells, e.g. certain b-cell-lymphomas. But the progress of this and many other techniques in the field of cell and gene therapy also depends on how well the sensitivity of the extracted organic material is dealt with.
Single Use Support has attended to this very challenge by providing dedicated protective shells for transporting and storing bioprocess containers, dealing with bags, vials and bottles as a primary packaging. The clear aim is to contribute to pioneering medical achievements from which a great number of patients shall profit.
FAQs about autologous and allogeneic therapies
What is the difference between autologous and allogeneic?
Autologous and allogeneic are terms used to describe the source of cells or tissues used for medical treatments or therapies. Autologous refers to cells or tissues that are obtained from the same individual who will receive the treatment, while allogeneic refers to cells or tissues that are obtained from a different individual of the same species. Autologous treatments may have lower risk of rejection, but may not be suitable for all patients due to limitations in the availability or quality of their own cells. Allogeneic treatments may have greater availability and diversity, but require matching and immunosuppressive measures to reduce the risk of rejection.
What are advantages and disadvantages of allogeneic?
Advantages of allogeneic treatments include a potentially wider availability and diversity of cells or tissues, while disadvantages include the risk of rejection and the need for matching and immunosuppressive measures to reduce this risk. Allogeneic treatments may also pose ethical and safety concerns, such as the risk of transmission of infections or diseases.
What are advantages and disadvantages of autologous?
Advantages of autologous treatments include a lower risk of rejection, as the cells or tissues are obtained from the same individual who will receive the treatment. Disadvantages include limitations in the availability and quality of the patient's own cells, which may not be suitable for all patients or conditions or result in reduced intensity. Autologous treatments may also be more expensive and time-consuming to produce.