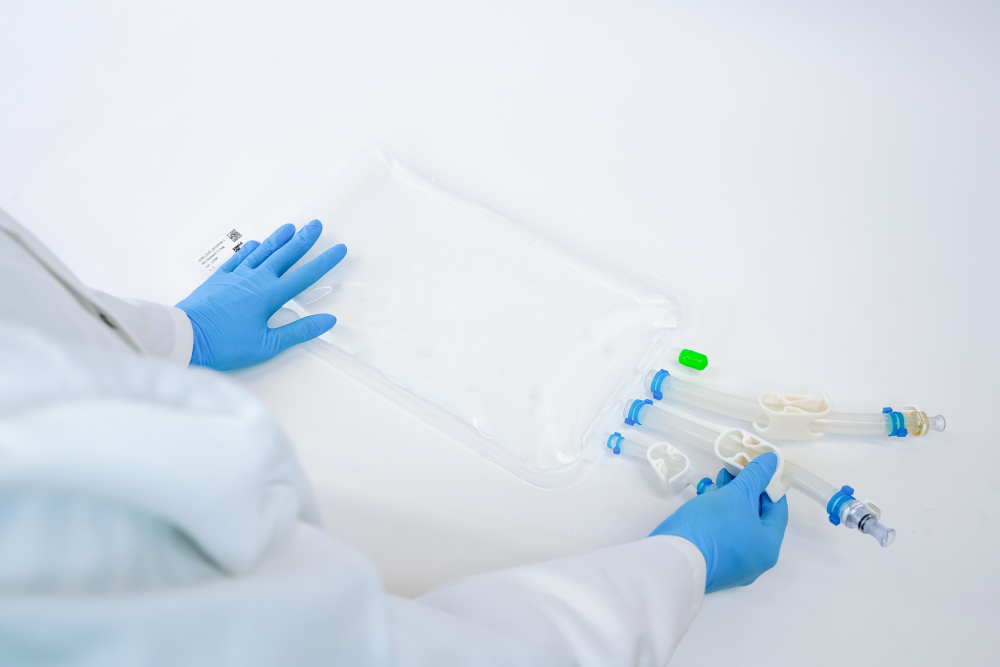
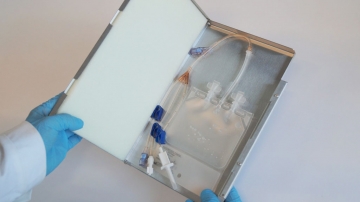
Our Single-Use Bioprocess Containers are fully qualified for the usage from -85°C to +55°C. Store, ship and freeze anything from cell culture samples to large volume drug substances. IRIS single-use bags are tailored to your needs.
IRIS: Innovative. Robust. Independent. Single-Use.