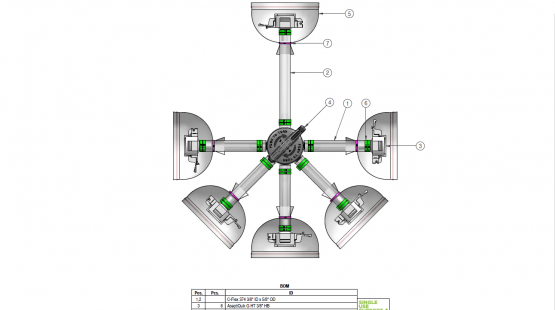
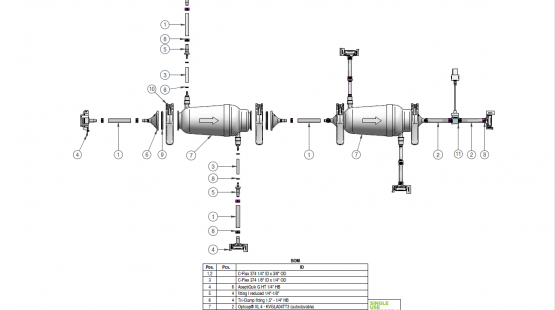
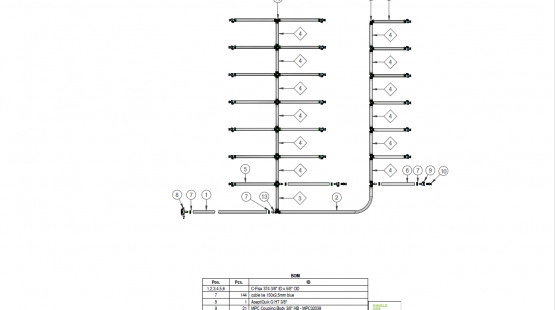
Single-use manifold assemblies are assembled single-use components that provide a sterile fluid flow of pharmaceutical products. We deliver manufacturer-independent single-use assemblies built to the highest quality standards in ISO 7 cleanrooms and sterilized within the shortest lead times.

Components used for IRIS Single-Use Assemblies
Single Use Support provides high quality, reliable and customized assemblies of tubing, connectors, fittings, primary packagings, beta bags, filters, and more. As experts in biopharmaceutical process solutions, we streamline your process by assisting you from component configuration to drawings to use.

DOWNLOAD WHITEPAPER
Whitepaper: Single-use assemblies for advanced fluid management
Throughout the entire manufacturing process, pharmaceutical liquids are transferred and have touchpoints within upstream bioprocessing, downstream bioprocessing and fill/finish. Single Use Support provides single-use assemblies of highest quality, reliability and speed of delivery. And with it capabilities in
- Dual Sourcing | Strengthening the Supply Chain
- Customization | Developing off-the-shelf assemblies
- Qualification | Providing GMP-compliant solutions
Our customers trust in us:
From component configuration to drawings to usage
First, the single-use assembly components must be defined and designed. Then, the required components are determined and the final drawing are added to the product drawing. Once approved the manufacturing is performed in ISO 7 cleanrooms, followed by labeling, packaging and sterilization.

Single-Use Assembly Catalogue
Design and configure your custom single-use assemblies according to individual needs and requirements. All our single-use components comply with any FDA, ISO and other regulatory standards and guarantee aseptic handling of your matter. After all, you want to avoid leachables, extractables or any kind of cross-contamination to impact your product before it reaches the end user.
Download more datasheets
Click here to download technical product data sheets, case studies and more.

Datasheet Single-Use Assemblies
Filesize: 0.7 MB – Mime-Type: application/pdf

Case Study: Single-use assembly capabilities to keep pace with biopharma dynamics
Filesize: 1.27 MB – Mime-Type: application/pdf

Whitepaper: Connecting the Dots - IRIS Single-Use Assemblies
Filesize: 0.7 MB – Mime-Type: application/pdf
FAQ about single-use assemblies
What is a single-use assembly?
A single-use assembly is a custom-made component for processing various goods, such as biologics or other products, that need to be kept germ-free.
Single-use assemblies are often made out of plastic and make sure that workflows are seamless and safe during production and processing, e.g. single-use filtration assemblies.
One great advantage of single-use assemblies is that they are sterile when firstly (and lastly) used and do not need to be cleaned for any additional use, which is both cost- and risk-minimizing.
What validation guidelines are in place at Single Use Support manifolds?
Single Use Support’s commitment to qualification standards means providing solutions that adhere to the stringent requirements of GMP, including Annex 1 guidelines. We assist clients in qualifying their processes with comprehensive documentation aligned with current Good Documentation Practices (cGDP).
The manufacturing of single-use assemblies is performed in ISO 6 cleanrooms, followed by labeling, packaging and sterilization.
Is it possible to customize single-use assemblies?
Yes, Single Use Support offers customized solutions, where CDMOs and manufacturers can develop single-use manifolds tailored to their specific fluid paths. Once customized, they can get their single-use assembly off-the-shelf, anytime.
What single-use components can be purchased via Single Use Support?
Anything you require can be included in your single-use manifold assembly. Be it equipment for filtration, clamps, gaskets, beta bags and stainless-steel protective shells for cell culture media, or other components based on single-use technology.
Are single-use filtration assemblies from Single Use Support compatible with other manufacturers?
IRIS Single-use filtration assemblies offer full flexibility since they are manufactuerer-independent. The filtration assemblies are easy to use with our RoSS.Fill platform system or any other platform from other suppliers.
Why should I implement a dual sourcing strategy?
Dual- or multiple sourcing strengthens the biopharmaceutical supply chain by providing a validated safety net, increasing flexibility, and promoting long-term stability, based on your preferred single-use components. It ensures that supply sources are well-managed, with contingency plans in place to handle unforeseen disruptions. For example, if a specific single-use component from Vendor X is unavailable, a Vendor Y alternative can be seamlessly integrated, maintaining production continuity.