Differences between microbial fermentation & mammalian cell culture
Table of contents
ShowMicrobial and mammalian cell fermentation are important processes in the development of media and drug development for the production of biologics, most importantly vaccines. However, there are certain characteristics that differentiate them from another in their use in bioengineering.
In this article, both methods of pharmaceutical fermentation and their differences will be explained. Moreover, it will be established for which production method in biomanufacturing they are more or less suited and why.
What is mammalian cell culture?
Mammalian cell culture describes the process of growing cells in-vitro, meaning outside their natural environment. For this cell culture process the animal cell is added to cell culture media inside a bioreactor or flask. Necessary instrumentation for the fermentation process is a bioreactor design that allows for controlled conditions to enhance cell growth like perfusion and monitoring of dissolved oxygen.
During the growth process mammalian cells, which are eukaryotic cells, use fermentation as a pathway to break down glucose. Glycolysis, as well as fermentation, both occur in the cytoplasm in eukaryotic cells. Mammalian cell lines like Human Embryonic Kidney cells (HEK cells) or Chinese Hamster Ovary cells (CHO cells) are used for biotechnology in antibody production, the production of hormones or enzymes and have led to the development of numerous biotherapeutics and biopharmaceuticals. Moreover, they are an important research tool in biology, cytotechnology, physiology and medicine.
What is microbial fermentation?
Microbial fermentation has its origin in food production. Thus, the production of alcoholic beverages like beer relying on alcoholic fermentation using yeast strain is one example of microbial metabolite production as well as the cultivation of sour dough bread. In contrast to mammalian cell culture, microbial fermentation is based on prokaryotic cells like lactate bacteria or yeast fungi.
However, other industries were able to benefit from microbial fermentation as well. It underwent a process development and is now used in medicine, microbiology and biotechnology for the production of biologicals like microbial enzymes, biomass, amino acids or recombinant protein. E. coli microbes have shown themselves to be very valuable in the production of biologic drug substances, while 1 Saccharomyces cerevisiae and Pichia pastoris are two kinds of yeast most commonly used for pharmaceutical manufacturing.1
Differences between the fermentation of a mammalian cell and a microbial cell
As we have already established, fermentation can occur in mammalian cells as well as microbial cells. While mammalian and microbial cell cultures are both methods to grow cells under controlled laboratory conditions and both rely on growth enhancing nutrients as impellers and the screening of aeration and oxygen transfer for quality assurance, there are key differences between the two.
As the name already suggests, one relies on the growth of mammalian cells, while the goal of the other is to grow and multiply microbes, microbial cells do not require a matrix to adhere. They are less complicated and expensive to work with than mammalian cells and are generally preferred in the manufacturing process of smaller biologics due to high production yields and shorter process times than those associated to mammalian cell culture. Moreover, they make it possible to produce smaller biologics on large-scale.2
Mammalian cell culture for the production of biologics
Mammalian cell culture is the most popular method for the production of biologics. For the expression of antibodies and other large proteins that require post-translational modifications, the pharmaceutical industry leans strongly towards the use of mammalian cell culture.
Only in 2018, almost 70 % of biologics were produced using mammalian cell culture, for the most part monoclonal antibodies, or mAbs, produced with the help of CHO cells which have a high functionality and good scale-up prospects as a host cell line.
Microbial fermentation in biomanufacturing
While mammalian cell culture is the preferred method for the production of antibodies and large proteins with post-translational modifications, their high-cost and long process-times caused interest in microbial fermentation to rise.
Microbial fermentation in bacteria, yeast or fungi is, due to its benefits, preferred in the production of smaller biologics. These include peptides, proteins, cytokines, growth factors, plasmid DNA, single-domain antibodies, peptibodies and non-glycosylated antibody fragments.3
Microbial fermentation vs mammalian cell fermentation: What's the better choice?
While mammalian cell culture has been the leading method in the production of antibodies and large proteins in recent years, their high production costs and long process-time characterization make microbial fermentation the method of choice in the production of smaller biologics.
Microbial fermentation is the better option in the production of complex drug substances like single-domain-antibodies, peptibodies, as well as antibody-fragments. The development times are shorter, production yields are higher, filtration is easier to achieve and quality variation between fed batches tends to be lower compared to mammalian cell culture.4
Fermentation manufacturing solutions
Current good manufacturing practices, short cGMP, of upstream bioprocessing rely on the assurance of product quality, controlled processes and safety. As the main goal in microbial fermentation is the optimization of microorganism cultivation while achieving high-cell density and promoting growth rate in the final culture generation, there are methods and precautions to be taken.
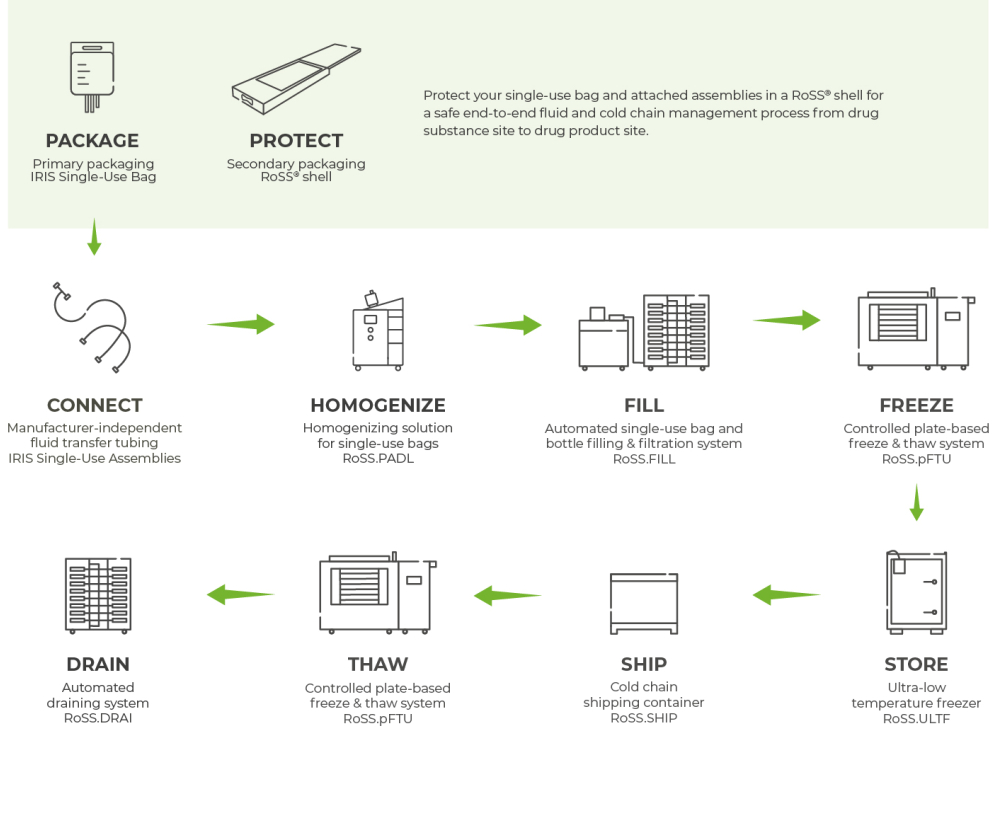
Especially if large quantities of process intermediates have to be transported for completion and final conjugation or manufacturing, quick freezing and thawing are most important. For this reason, Single Use Support offers flexible end-to-end process solutions for advanced fluid management and freeze-thaw logistics involving single-use technologies. These include fast aseptic and fully automated aliquotation into single-use containers, as well as controlled freezing methods up to - 80°C in less than eight hours.
- Microbes of traditional fermentation processes as synthetic biology chassis to tackle future food challenges, http://dx.doi.org/10.3389/fbioe.2022.982975, Published 2022-09-16
- A Review of the Microbial Production of Bioactive Natural Products and Biologics, http://dx.doi.org/10.3389/fmicb.2019.01404, Published 2019-06-20
- F. Mirasol. “Weighing the Benefits of Fermentation for New Biotherapies,” BioPharm International 35 (10) ., https://, Published 18–22 (2022)
- Solid-state fermentation: a promising microbial technology for secondary metabolite production, http://dx.doi.org/10.1007/s002530000565, Published 2003-02-13










